

FEATURED BLOG

Why Cloudbyz CTMS is the Best Choice for Biotechnology Companies
For biotechnology companies looking to streamline their clinical trial management, reduce time to market, and achieve better outcomes, Cloudbyz CTMS is the solution that delivers. Its specialized features, scalability, and commitment to innovation make it the best choice for managing the complexities of biotechnology clinical trials today and in the future.

Why Cloudbyz CTMS is the Best Choice for Biotechnology Companies
For biotechnology companies looking to streamline their clinical trial management, reduce time to market, and achieve better outcomes, Cloudbyz CTMS is the solution that delivers. Its specialized features, scalability, and commitment to innovation make it the best choice for managing the complexities of biotechnology clinical trials today and in the future.

FEATURED BLOG
September 25, 2020 by Ani Mayasandra
We analyzed 164 pharmacovigilance startups impacting the industry. HEPAprint, MEDIKURA, Navro, Embleema & Cloudbyz develop 5 top solutions to watch out for. Learn more in our Global Startup Heat Map!
Blog > All

Comparing the Top Clinical Trial Management Systems (CTMS) in 2024
As the clinical research landscape becomes increasingly complex and data-driven,

Harnessing the Power of Real-World Evidence: How Cloudbyz eClinical Suite Enables Comprehensive RWE Integration
With Cloudbyz, companies can confidently navigate the complexities of RWE, transforming data into actionable insights that improve patient outcomes, accelerate drug development, and support regulatory decisions. The future of clinical research is here, and with Cloudbyz eClinical suite, your organization can be at the forefront of this exciting transformation.

The Future of Clinical Operations: Why Cloudbyz eClinical Platform Outshines Standalone Solutions for Biotech Companies
As the biotech industry continues to evolve, the need for a comprehensive, integrated approach to clinical trial management becomes increasingly clear. While standalone solutions like Castor EDC and ClinCapture have their place, they simply cannot match the advantages offered by a unified platform like Cloudbyz eClinical. By providing seamless integration, comprehensive oversight, scalability, and a superior user experience, Cloudbyz empowers biotech companies to overcome the challenges of modern clinical research and accelerate the development of life-saving therapies. In an industry where time and accuracy are paramount, the choice is clear—Cloudbyz eClinical is the better solution for biotech companies looking to stay ahead of the curve.

How AI Can Enhance Clinical Data Management
Cloudbyz EDC is a powerful tool that enhances the quality, compliance, and efficiency of clinical trials. Its user-friendly interfaces, robust collaboration features, and comprehensive data security measures make it an ideal choice for organizations seeking to streamline their clinical research processes. By leveraging Cloudbyz EDC, researchers can ensure that their data is accurate, compliant, and secure, ultimately contributing to the success of their clinical trials.

Ensuring Compliance with Health Canada’s Foreign Actions Profile: A Digital Solution
Navigating the complexities of Health Canada’s Foreign Actions Profile requirements necessitates a proactive and integrated approach. By leveraging digital solutions, pharmaceutical companies can streamline their compliance processes, reduce the risk of non-compliance, and ensure the safety and efficacy of their products in the Canadian market. Embracing these technologies not only enhances regulatory adherence but also promotes operational efficiency and risk management, positioning companies for success in an increasingly regulated global environment.

Mergers in Pharma and Biotech: Shaping the Future of Clinical Trials Technology
The wave of mergers and acquisitions in the pharmaceutical and biotech sectors is reshaping the clinical trials landscape, driving substantial investments in advanced management technologies. These investments are crucial in addressing the complexities of modern clinical trials, enhancing efficiency, ensuring regulatory compliance, and ultimately accelerating the development of life-saving therapies.

Transforming Patient Support and Access Programs in Pharmaceutical Companies with Salesforce Health Cloud
In an era where patients are increasingly taking an active role in managing their healthcare, pharmaceutical companies must adapt by offering patient-centric support and access programs. Salesforce Health Cloud provides the tools and capabilities necessary to transform patient support and access, enabling pharmaceutical companies to deliver personalized care, engage with patients effectively, and streamline access initiatives. Embracing this CRM platform can not only improve patient outcomes but also enhance the reputation and competitiveness of pharmaceutical companies in the evolving healthcare landscape.

Understanding Electronic Data Capture (EDC) in Clinical Research
Electronic Data Capture systems are transforming the way clinical trials are conducted, offering significant improvements in data accuracy, management, and compliance. By carefully evaluating the key capabilities and choosing a reliable provider, organizations can leverage EDC systems to enhance the efficiency and success of their clinical research efforts. EDC is not just a technological advancement; it is a critical component of modern clinical trials that drives better outcomes and fosters innovation in medical research.
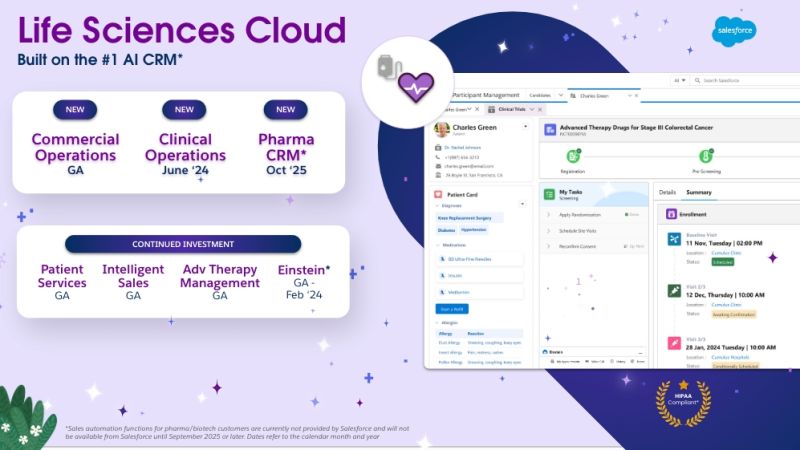
How Cereblis Can Elevate the Potential of Salesforce’s New Life Sciences Cloud Launch for Clients
Salesforce’s Life Sciences Cloud is set to be a game-changer for the industry. But like any tool, its success in a company hinges on its implementation and adoption. With Cereblis’s expertise in both Salesforce and the life sciences sector, clients are assured of a partner who understands their challenges and can guide them to success in this new digital era.

25 Reasons Why Pharmaceutical Companies Need a Unified Clinical Trial Management Platform Built on Salesforce
A unified clinical trial management platform built on Salesforce offers a versatile and powerful solution for pharmaceutical companies. With its scalability, regulatory compliance, and robust features, Salesforce empowers organizations to streamline their clinical trial operations, reduce costs, and accelerate drug development. By embracing this platform, pharmaceutical companies can not only enhance their efficiency but also contribute to the timely delivery of life-saving medications to patients worldwide.

Best Practices in Market Access Programs for Pharmaceutical Companies
Market access is a multifaceted challenge for pharmaceutical companies. By adopting these best practices, companies can navigate the complex landscape effectively, ensure timely patient access to their products, and achieve commercial success while making a positive impact on patient outcomes. Effective market access programs are not only essential for business success but also for improving healthcare systems and patient care worldwide.

Investigator Initiated Trials (IIT), Investigator Initiated Research (IIR), and Grants Management: Digitization with Salesforce Experience Cloud
Investigator Initiated Trials (IIT), Investigator Initiated Research (IIR), and Grants Management are vital components of advancing medical research. Salesforce Experience Cloud offers a comprehensive solution for digitizing and streamlining these processes, ensuring efficient management, compliance, and collaboration among stakeholders. By harnessing the power of Salesforce Experience Cloud, research institutions can accelerate the pace of discovery and make a meaningful impact on patient care and scientific knowledge.
Cereblis enables companies to take advantage of innovation in tech by onboarding new ways to work and execute projects by advising, implementing, and optimizing digital solutions.
Cereblis LLC, 1770, Park Street,
Suite 108, Naperville IL 60563.
Phone: +1 (312)-763-8040
Email: info@cereblis.com